What we do
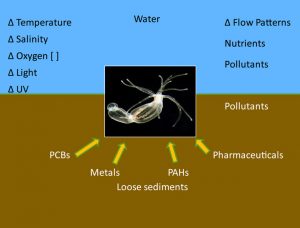
We use molecular tools to better understand how animals detect and respond to signals and stresses in the marine environment.
For example, in temperate areas, we experience strong daily and seasonal cycles in temperature and light. As animals ourselves (!), we respond to these cycles. Most of us sleep at night, work and play during the day, wear mittens and scarves in the winter, and change into short sleeves in the summer. Beyond these behavioral changes, our physiology changes. Our metabolic machinery is prepared for us to start eating breakfast in the morning. As July and August approach, we build up a larger blood volume that allows us to sweat and cool ourselves better. Similarly, marine animals have evolved diverse strategies for coping with a changing world…whether in response to physical conditions, ecological interactions, or human influences.