This being my first Alvin cruise, I had no idea what to expect. Well, Alvin is down in the water during the day (7am to 5pm), meaning any other shipboard sampling has to take place at night. The de facto result is that all the water casts occur at night. That’s not inherently a problem, but the crux of activity revolves around Alvin and so the few of us sampling water pretty much have the ship to ourselves. Not wanting to miss the bustle of activity associated with the dives (not to mention the celebratory return of each new diver), we also very much want to be up during the day too. Oh, and that whole science thing where we’re able to learn about what happened during the dives and look at a vast array of photos, etc., pretty much necessitates being up during “normal working hours.” I’m not complaining, though; far from it. I love research cruises! The camaraderie, the excitement, and the curiosity of how the world works around us all make for an astounding adventure. We’re occupying our days running the water column samples for nitrite and nitrate concentrations, and doing miscellaneous tests to make more precise measurements. We’re also helping others to get their science done. All in all, a good time.

Ryan Woosley measuring nitrite with a miniaturized spectrophotometer.
Nitrite is measured using a spectrophotometric assay. That’s fancy speak for: “we add some chemicals and manufacture a colored dye whose intensity is proportional to how much nitrite is in the sample.” A few years back, I did these measurements on a cruise in the North Atlantic from Rhode Island to Iceland with my twin sister, and we authored a blog for her high school chemistry students (she’s an awesome science teacher for those who aren’t aware). But, since the internet on this boat isn’t the greatest to copy and paste the full chemistry, I’ll just summarize how it works. I very much encourage you to go to northatlcruise.wordpress.com to see the full chemistry. The reaction is rather simple: we’re using organic chemistry to synthesize an azo-dye from the nitrite. More nitrite = more dye = more intense color (it’s magenta!). So, we take our seawater sample, add Sulfanilamide and N-1napthylethylenediamine dihydrochloride (we call this second one NED for obvious reasons), and wait a few minutes for the color to develop. Because the solution turns magenta, it absorbs green light (we don’t see the green reflected), and we can use a spectrophotometer to measure how much of the green light we shine on the sample gets absorbed. In summary: amount of nitrite in a sample relates to how much azo-dye is formed, which in turn relates to how much green light is absorbed. All said, we can then use the absorbance to calculate how much nitrite was in the sample in the first place!
Nitrate is a bit more tricky of a method to describe, so I think I’ll wait for a later blog post (it really deserves its own dedicated description). That reminds me… I should stop writing this post and start analyzing some samples. Stop distracting me!
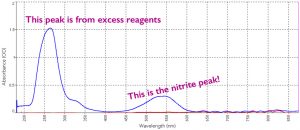
Spectrogram of a nitrite sample, showing how much of each wavelength of light is absorbed. The two peaks indicate absorbance in the ultraviolet (the left peak) by excess reagents and in the green (the right peak) by nitrite. Each color of light has a certain wavelength, and whatever is absorbed, you don’t see (it can’t reach your eye if a sample absorbed it!). So something that absorbs in the green appears magenta because it reflects blue and red light.

Nitrite samples, with increasing concentrations from left to right. The amount of color relates directly to how much nitrite is in the sample.


